Prenatal Stress and Child Development: Translating Animal Studies to Human Health
Thomas G. O’Connor, PhD
University of Rochester Medical Center, USA
Introduction
Prenatal stress is a leading paradigm used in experimental animal studies to demonstrate that early stress exposure can have lasting impact on the behaviour and biology of the offspring.1 If the strength, persistence, and diversity of effects of prenatal stress from these studies extend to humans, then the implications for public health and prevention are enormous. Accordingly, research on stress in human pregnancies has burgeoned in recent years, and this now constitutes a major research area on several continents.
Subject
A model underlying research on prenatal stress in humans has been discussed by several groups.2-3 The starting point is that the fetal period is one of particular ontogenetic vulnerability. That is, exposures – good or bad – during this time would be expected to have lasting effects. Even casual observers of science and medicine would be familiar with how certain molecules, such as folic acid, could have a dramatic influence on the long-term health outcomes of the developing fetus. The prenatal stress model makes a parallel hypothesis focusing on stress hormone exposure, and cortisol in particular – although there may be a number of mechanisms involved. Cortisol is a downstream product of the Hypothalamic-Pituitary-Adrenal (HPA) axis, one of the most-studied mind-body systems. Research has focused on the HPA axis for several reasons. One is that there are human data showing that cortisol crosses the placenta.4 Although the placenta screens out many substances, and there is an enzyme (11BHSD2) that prevents the crossing of cortisol, but it does so imperfectly. That implies that stressed or anxious mothers who have elevated cortisol may have fetuses who are exposed to elevated levels of cortisol in utero. If prenatal maternal anxiety or stress did alter fetal HPA axis via early cortisol exposure, then the implications for development would be substantial. That is because of the wide reaching impact of the HPA axis on many areas of functioning, including stress physiology, cognition and memory, immunology and cardiovascular health.5
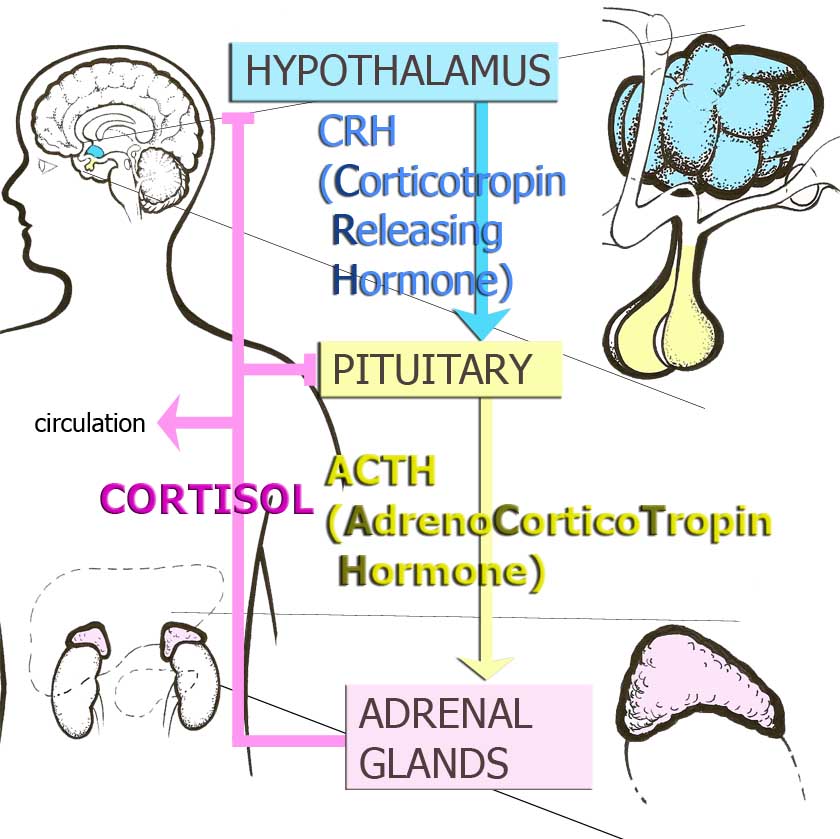
Figure 1. Hypothalamic-Pituitary Adrenocortical Axis or Stress Hormone Axis.
Problems
The number of studies linking stress or anxiety to child outcomes is impressive and mostly consistent, but uncertainties remain about the mechanisms involved. Additionally, most of the studies included largely middle-class samples; whether or not the effects of prenatal stress extend to high-risk samples, including samples from developing countries, is unknown and represents a notable gap in the literature.
Research Context
Research on prenatal stress in humans follows an established paradigm in experimental animal work dating back many decades. The research context of the animal work is especially compelling, but the translation to human development is incomplete. That is partly because the precisely-timed, well-controlled nature of stress exposure used in most animal studies does not easily apply to the kinds of chronic stress-exposed families of most interest to clinicians and social policy-makers.
Key Research Questions
Findings emerging from studies in the U.S., Canada, Europe and Australia all suggest that there is an association between prenatal anxiety or stress and a range of child development outcomes. The questions for research are now:
- What are the mechanisms by which prenatal stress or anxiety causes (if it is causal) a range of biobehavioural outcomes in the child?
- Are the effects of prenatal stress or anxiety modifiable by intervention in pregnancy or the postnatal period, or by the early caregiving environment?
- Is it practical to identify which children may be at risk based on a prenatal screening of the mother?
Recent Research Results
Research groups in several countries link prenatal stress or anxiety to a range of child outcomes. The more reliable links are with lower cognitive and language ability;6 more difficult, inhibited, or emotionally reactive temperament;7 behavioural problems;8 and poorer neurodevelopmental outcomes.9 Studies linking prenatal stress or anxiety to child outcomes have extended the longitudinal follow-up to adolescence.10
Significantly, the push to examine the prenatal stress/anxiety hypothesis often missed another key element, the role of postnatal rearing. Experimental animal studies demonstrated that the effects of prenatal stress could be eliminated by positive postnatal rearing.11 Unfortunately, with few exceptions, human research on prenatal anxiety or stress has ignored the early caregiving environment. One exception is a recent study12 that found that elevated levels of cortisol in amniotic fluid, taken at an average age of 17 weeks gestation, predicted lower cognitive ability in the infant. More impressive was that this association was entirely dependent on the quality of the child-parent relationship: for children who experienced less than optimal care, amniotic fluid cortisol strongly negatively predicted infant cognitive ability; however, for children who experienced a sensitive-response caregiving environment, there was no association between amniotic fluid cortisol and infant cognitive ability.
Research Gaps
Two main gaps remain in the human research on prenatal stress and anxiety and child outcome. The first is confirmation of a mechanism or mechanisms. Cortisol, as noted, is the leading candidate mechanism, but research has yet to show that it clearly mediates the effects of prenatal maternal stress on child outcomes; indeed, evidence of non-mediation has been reported.12 In addition, it is essential not to see cortisol as an inevitably adverse exposure. Cortisol, and glucocorticoids more generally, have numerous biological functions, including a functional role in childbirth, for example. That is, it is important to understand cortisol as both serving essential, functional and potential adverse roles in human development – a tricky balance that is not yet firmly rooted in how research is conducted.
A second major gap concerns intervention. One might speculate that reducing anxiety or stress during pregnancy through an array of psychological treatments might prevent the adverse effects in the child. Unfortunately, no clear evidence yet exists. Randomized controlled trials that capitalize of the leverage of a treatment study would provide the most powerful demonstration of a causal effect and offer concrete clinical guidance for those caring for stressed and anxious pregnant mothers. It is unfortunate that so much of the attention on treatment focuses on psychiatric medications. Psychological treatments such as cognitive behaviour therapies are effective and do not carry iatrogenic risk.
Conclusions
Prenatal maternal stress or anxiety has been shown in a variety of studies to be a risk for poor or compromised development in the child, indexed by such measures as cognitive and language ability, temperament, neurodevelopment, and behavioural and social adjustment. The weight of the evidence suggests that this is rightly seen as a potential clinical and public health concern that warrants particular attention. No causal link can yet be drawn due to the limited leverage of existing human studies. Much of the speculation about mechanisms focuses on the HPA axis and its downstream product cortisol. One important benefit for research is that cortisol can be measured from saliva, making it an accessible – if not fully understood – biological marker of stress in the mother and risk to the child. One of the more notable findings from recent studies is that early parent-child relationship quality may eliminate or otherwise modify the effects of prenatal stress on the child. That raises implications for intervention that are only now being investigated.
Implications for Parents, Services and Policy
The identification of causal mechanism is important for scientists for advancing theories and biological models and for clinicians whose construction of intervention relies on a clear and sound understanding of how an intervention works. Policy-makers are equally dependent on understanding mechanisms so that money is not wasted on mis-targeted programs. The case for parental understanding of mechanisms – to understand better their children's development – is no less subtle. Thus, curiosity about why prenatal stress may pose a risk is not merely an academic concern.
A more obvious implication is prevention. If it were possible to prevent the adverse effects of maternal prenatal stress or anxiety on the child – and this has not yet been soundly demonstrated – then policies could be put in place to cost-effectively promote the well-being of women and children. Several outcomes linked to prenatal stress or anxiety, namely, low birthweight, prematurity and neurodevelopmental problems in children carry sizable costs and prevention efforts have not been very successful. It is not certain that reduction to maternal prenatal stress or anxiety would reduce the likelihood of these outcomes, but even the possibility should instigate systematic efforts and clinical trials. Work of this kind would have a positive side-effect of directly identifying which of the many programs promoted for pregnant women (mostly without an evidence base) actually benefit the mother or the child.
References
- Schneider ML, Moore CF, Kraemer GW, Roberts AD, DeJesus OT. The impact of prenatal stress, fetal alcohol exposure, or both on development: perspectives from a primate model. Psychoneuroendocrinology 2002;27(1-2):285-298.
- O'Donnell K, O'Connor TG, Glover V. Prenatal stress and neurodevelopment of the child: focus on the HPA axis and role of the placenta. Developmental Neuroscience 2009;31(4):285-292.
- Wadhwa PD, Sandman CA, Porto M, Dunkel-Schetter C, Garite TJ. The association between prenatal stress and infant birth weight and gestational age at birth: a prospective investigation. American Journal of Obstetrics & Gynecology 1993;169(4):858-865.
- Sarkar P, Bergman K, Fisk NM, O'Connor TG, Glover V. Ontogeny of foetal exposure to maternal cortisol using midtrimester amniotic fluid as a biomarker. Clinical Endocrinology 2007;66(5):636-640.
- Kapoor A, Matthews SG. Prenatal stress modifies behavior and hypothalamic-pituitary-adrenal function in female guinea pig offspring: effects of timing of prenatal stress and stage of reproductive cycle. Endocrinology 2008;149(12):6406-6415.
- Laplante DP, Brunet A, Schmitz N, Ciampi A, King S. Project Ice Storm: prenatal maternal stress affects cognitive and linguistic functioning in 5½-year-old children. Journal of the American Academy of Child and Adolescent Psychiatry 2008;47(9):1063-1072.
- Davis EP, Glynn LM, Schetter CD, Hobel C, Chicz-Demet A, Sandman CA. Prenatal exposure to maternal depression and cortisol influences infant temperament. Journal of the American Academy of Child and Adolescent Psychiatry 2007;46(6):737-746.
- O'Connor TG, Heron J, Golding J, Beveridge M, Glover V. Maternal antenatal anxiety and children's behavioural/emotional problems at 4 years. Report from the Avon Longitudinal Study of Parents and Children. British Journal of Psychiatry 2002;180:502-508.
- Obel C, Hedegaard M, Henriksen TB, Secher NJ, Olsen J. Psychological factors in pregnancy and mixed-handedness in the offspring. Developmental Medicine & Child Neurology 2003;45(8):557-561.
- van den Bergh BR, Marcoen A. High antenatal maternal anxiety is related to ADHD symptoms, externalizing problems, and anxiety in 8- and 9-year-olds. Child Development 2004;75(4):1085-1097.
- Maccari S, Piazza PV, Kabbaj M, Barbazanges A, Simon H, Le Moal M. Adoption reverses the long-term impairment in glucocorticoid feedback induced by prenatal stress. Journal of Neuroscience 1995;15(1 Pt 1):110-116.
- Bergman K, Sarkar P, Glover V, O'Connor TG. Maternal prenatal cortisol and infant cognitive development: moderation by infant-mother attachment. Biological Psychiatry 2010;67(11):1026-1032.
How to cite this article:
O’Connor TG. Prenatal Stress and Child Development: Translating Animal Studies to Human Health. In: Tremblay RE, Boivin M, Peters RDeV, eds. Glover V, topic ed. Encyclopedia on Early Childhood Development [online]. https://www.child-encyclopedia.com/stress-and-pregnancy-prenatal-and-perinatal/according-experts/prenatal-stress-and-child-development. Published: January 2011. Accessed June 5, 2026.
Text copied to the clipboard ✓